Call: 08045479200
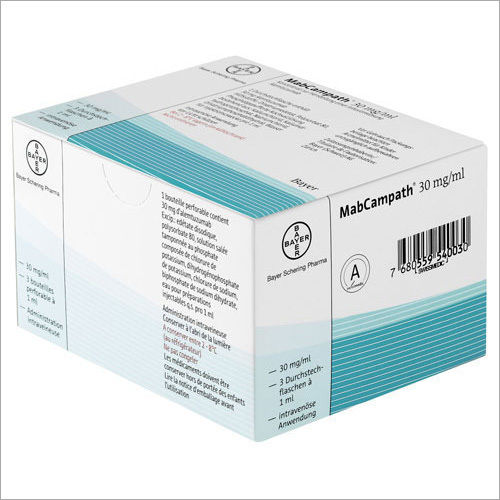
Mabcampath
Product Details:
- Indication Chronic Lymphocytic Leukemia (CLL)
- Salt Composition Alemtuzumab
- Dosage Form Injection
- Enzyme Types Monoclonal Antibody
- Feature Humanized monoclonal antibody for targeted cancer therapy
- Ingredients Alemtuzumab (humanized monoclonal antibody)
- Application Treatment of B-cell chronic lymphocytic leukemia
- Click to View more
X
Mabcampath Price And Quantity
Mabcampath Product Specifications
- Neutral (approx. pH 7.0)
- Alemtuzumab (humanized monoclonal antibody)
- Chronic Lymphocytic Leukemia (CLL)
- Clear, colorless solution
- Alemtuzumab
- Humanized monoclonal antibody for targeted cancer therapy
- Treatment of B-cell chronic lymphocytic leukemia
- Monoclonal Antibody
- Store between 2C and 8C. Do not freeze.
- 24 months
- Odorless
- Injection
Mabcampath Trade Information
- 500 , Per Month
- 7 Days
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
- All India
Product Description
MabCampath is used to treat patients with B-cell chronic lymphocytic leukaemia (B-CLL), a cancer of a type of white blood cell called B lymphocytes. MabCampath is used in patients for whom treatment combinations including fludarabine (another medicine used in leukaemia) are not appropriate. The medicine can only be obtained with a prescription.- Medicine Name: Mabcampath
- Generic Name: Alemtuzumab
- Approval Date: 2001
- Company Name: Genzyme Corporation
- Available as (Form & Strength): 30 mg/1 mL single use vial
Targeted Oncology Therapy for CLL
Mabcampath offers a focused treatment approach by leveraging the monoclonal antibody Alemtuzumab, designed specifically to recognize and eliminate B cells affected by chronic lymphocytic leukemia. This targeted action supports improved efficacy and limits unnecessary exposure to healthy tissues, leading to more precise cancer care for eligible patients.
Prescription Strength and Quality Assurance
Quality and safety remain paramount with Mabcampath. Each vial is GMP certified and formulated to pharmaceutical standards, ensuring a sterile, stable solution for intravenous administration. With strict prescription requirements and comprehensive storage guidelines, this product ensures both optimal potency and patient safety throughout its shelf life.
FAQs of Mabcampath:
Q: How should Mabcampath be administered to patients?
A: Mabcampath must be administered as an intravenous injection under direct supervision of a qualified healthcare professional experienced in oncology treatments. Dosage and administration schedules are determined by the prescribing physician based on individual patient requirements.Q: What are the primary benefits of using Mabcampath for chronic lymphocytic leukemia?
A: Mabcampath provides targeted therapy by specifically binding to CD52 antigens on B lymphocytes, leading to effective elimination of malignant cells while minimizing adverse effects on healthy tissues. This results in improved treatment outcomes for those diagnosed with B-cell chronic lymphocytic leukemia.Q: When is treatment with Mabcampath indicated?
A: Mabcampath is indicated for patients diagnosed with B-cell chronic lymphocytic leukemia, particularly when alternative therapies are unsuitable or have proven ineffective. Its use should be determined and monitored by an oncologist based on comprehensive clinical evaluation.Q: Where should Mabcampath be stored prior to use?
A: The vials should be stored at a temperature range between 2C and 8C, in a refrigerator. Freezing must be strictly avoided to maintain the efficacy and stability of the product throughout its shelf life.Q: What patients should not receive Mabcampath?
A: Mabcampath is contraindicated in patients with active infections. A thorough assessment should be conducted before initiation to rule out any ongoing infections, as the treatment can further weaken the immune system.Q: What quality and safety standards does Mabcampath adhere to?
A: Mabcampath complies with Good Manufacturing Practice (GMP) certification, ensuring its quality, sterility, and safety for intravenous administration as an oncology therapeutic. Every vial is produced under strict controls to guarantee patient safety.Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
Other Products in 'Anti Cancer Capsules' category
We Mainly Deal in International Market.
 Send Inquiry
Send Inquiry