Call: 08045479200
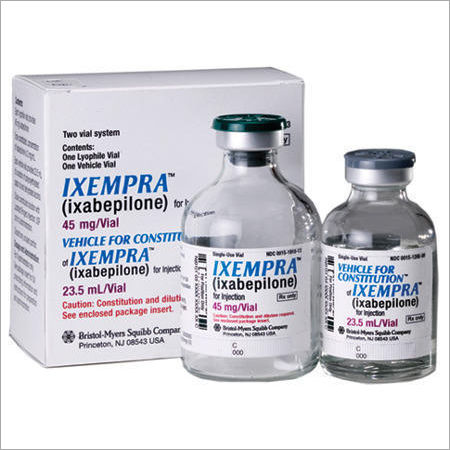
Ixempra Medicines
Product Details:
- Indication Breast Cancer treatment, particularly metastatic or locally advanced
- Dosage Form Injection
- Salt Composition Ixabepilone
- Feature Cytotoxic agent, ready to use injection
- Ingredients Ixabepilone, excipients
- Application Oncology/Anticancer therapy
- Physical Color/Texture Clear, Colorless to pale yellow liquid
- Click to View more
X
Ixempra Medicines Price And Quantity
- Bristol-Myers Squibb
- Yes
- 219989-36-9
- Antineoplastic
- For hospital use only, cytotoxic handling needed
- WHO-GMP Certified
- Intravenous
- Hypersensitivity to Ixabepilone or formulation excipients
- Vial
- 15 mg, 45 mg per vial
- L01D
Ixempra Medicines Product Specifications
- Ixabepilone
- 2 years from manufacture
- Clear, Colorless to pale yellow liquid
- Cytotoxic agent, ready to use injection
- Store at 2C to 8C (Refrigerate), Protect from light
- Oncology/Anticancer therapy
- Ixabepilone, excipients
- Injection
- Breast Cancer treatment, particularly metastatic or locally advanced
- Bristol-Myers Squibb
- Yes
- 219989-36-9
- Antineoplastic
- For hospital use only, cytotoxic handling needed
- WHO-GMP Certified
- Intravenous
- Hypersensitivity to Ixabepilone or formulation excipients
- Vial
- 15 mg, 45 mg per vial
- L01D
Ixempra Medicines Trade Information
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
Product Description
These medicines belong to class of medications called antineoplastic agents. We have been striving to maintain high quality standards in the market by offering Ixempra Medicines. It is used in hospitals and nursing homes for the treatment of advanced breast cancer. We procure these medicines with the help of our agents from reliable vendors. Ixempra Medicines work by preventing abnormal protein activity which signals cancer cells to multiply and spread across the body.
Features:
- Slow down cancer cells
- Highly effective anti cancer drug
- Extended shelf life
Specifications:
- Available in: 45 mg
Targeted Anticancer Therapy for Breast Cancer
Ixempra is specifically developed for patients with metastatic or locally advanced breast cancer who may not respond to other treatments. The active ingredient, Ixabepilone, disrupts cancer cell growth, offering a valuable option in oncology. Its use is restricted to healthcare professionals experienced in handling cytotoxic medications, ensuring optimal efficacy and safety in a hospital environment.
Stringent Manufacturing and Handling Standards
Manufactured under WHO-GMP guidelines and marketed by Bristol-Myers Squibb, Ixempra ensures quality and safety throughout its lifecycle. This cytotoxic injection should only be administered intravenously by trained medical personnel, following proper preparation, storage, and disposal protocols. For best results, it must be stored refrigerated (28C) and protected from light.
FAQs of Ixempra Medicines:
Q: How is Ixempra injection administered to patients?
A: Ixempra is administered as an intravenous injection by a qualified healthcare professional in a hospital setting. The dosage and schedule are determined by the oncologist, tailored to each patients needs.Q: What are the primary indications for Ixempra use?
A: Ixempra is indicated for the treatment of metastatic or locally advanced breast cancer, particularly in patients who have not responded to certain other cancer therapies.Q: When should Ixempra not be used?
A: Ixempra is contraindicated in individuals with hypersensitivity to Ixabepilone or to any excipient in the formulation. Always inform your doctor about any allergies before starting treatment.Q: Where should Ixempra be stored before use?
A: The vials of Ixempra should be kept in a refrigerator at 2C to 8C and protected from light. Improper storage can impact the medicines efficacy and safety.Q: What is the process for handling and disposing of Ixempra vials?
A: As a cytotoxic agent, Ixempra requires careful handling by trained staff in accordance with hospital protocols. Proper protective equipment must be used, and any unused product or waste materials should be disposed of as per local cytotoxic waste guidelines.Q: What benefits does Ixempra offer in oncology treatment?
A: Ixempra provides an effective option for patients with advanced breast cancer, especially those unresponsive to other therapies. Its valued for its unique mode of action and availability in ready-to-use vial form, streamlining treatment in hospitals.Q: How can I obtain Ixempra for cancer therapy?
A: Ixempra is a prescription-only medicine distributed, exported, and traded by authorized parties in India. It is available to hospitals and healthcare professionals for use in accordance with oncology treatment protocols.Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
Other Products in 'Anti Cancer Injections' category
We Mainly Deal in International Market.
 Send Inquiry
Send Inquiry
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese